 Its mass is almost the same as that of the proton. As its name implies, this particle carries no electrical charge. The other nuclear particle is the neutron. The symbol Z comes from the German word zahl, which means numeral, or atomzahl. It is denoted by the symbol Z and is the subscript in atomic notation. 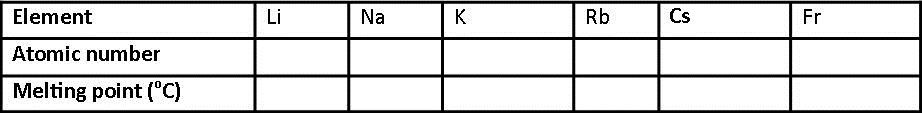
The atomic number is also called the proton number. 
This means that in any atom, the number of protons in the nucleus (often referred to as the nuclear charge) is balanced by the same number of electrons outside the nucleus. The atomic number is the number of protons found in the nucleus of an atom, which uniquely identifies its element. Protons are the carriers of positive electric charge in the nucleus the proton charge is exactly the same as the electron charge, but of opposite sign. The nucleus is itself composed of two kinds of particles. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. Element Silver (Ag), Group 11, Atomic Number 47, d-block, Mass 107.868. The radius of an atom must be defined arbitrarily, such as the boundary in which the electron can be found with 95% probability. Periodic Table of Videos Created by video journalist Brady Haran working with chemists at The University of Nottingham. The latter has no definite outer boundary, so neither does the atom. It is common (but somewhat misleading) to describe the volume of space in which the electrons of an atom have a significant probability of being found as the electron cloud. The extremely small mass of the electron (1/1840 th the mass of the hydrogen nucleus) causes it to behave as a quantum particle, which means that its location at any moment cannot be specified the best we can do is describe its behavior in terms of the probability of its manifesting itself at any point in space. 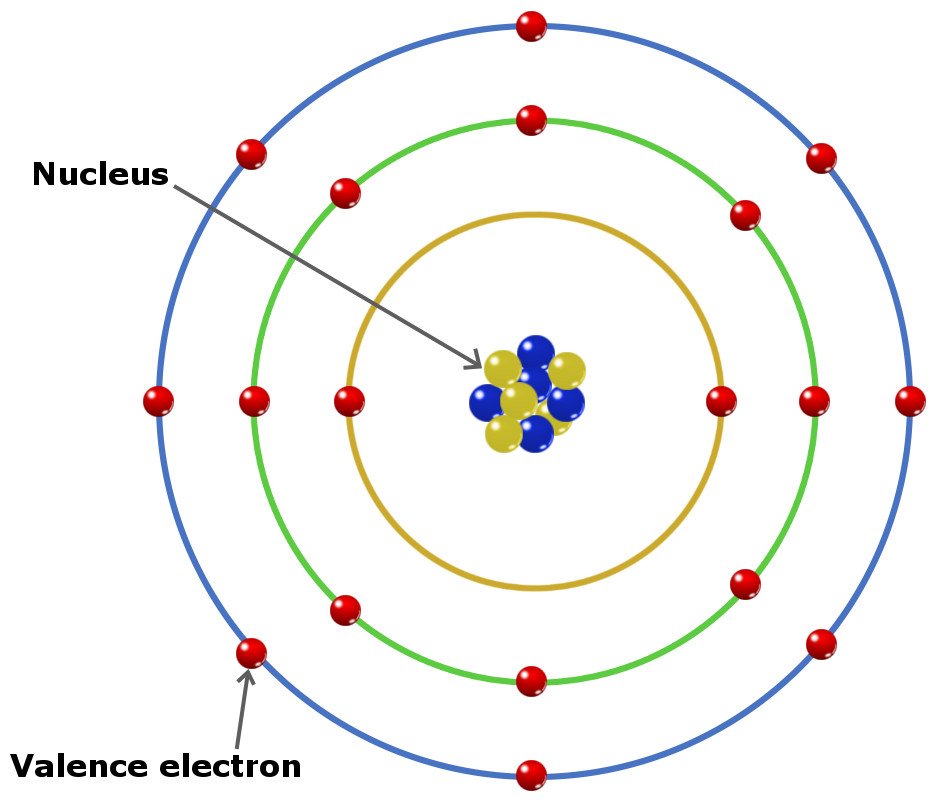
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
